Neural tube defects, including Spina bifida, are among the most common congenital disorders, affecting approximately one in 1,000 live births worldwide. These conditions are linked to the process of junctional neurulation. Although neural tube defects typically develop within the first four weeks of pregnancy, observing the specific molecular mechanisms behind their formation has historically been challenging. A research team at the University of Queensland has overcome this hurdle by employing sophisticated imaging technologies and transgenic quail embryos to observe, in real time, how the PRICKLE1 protein regulates cellular events during neural tube formation. This new knowledge, obtained by tracking PRICKLE1 activity in junctional neural tube defects, paves the way for innovative approaches to clinical diagnosis and preventive treatment.
Understanding the Role of PRICKLE1 in Human Birth Defects
PRICKLE1 is a critical protein involved in cellular development, particularly in the formation of tissue structures. For many years, it has been studied for its role in planar cell polarity (PCP) signaling, which helps cells orient themselves. However, new research reveals that PRICKLE1 has another function, independent of the PCP pathway, in the formation of the junctional neural tube. According to a thesis from the University of Queensland, PRICKLE1 assists in assembling the cortical actomyosin network of midline cells, a process essential for apical constriction. This provides the driving force for cell movement during epithelial-mesenchymal transition (EMT). When PRICKLE1 is disrupted, cellular ingression into the neural tube is impaired, leading to structural defects in the spinal cord.
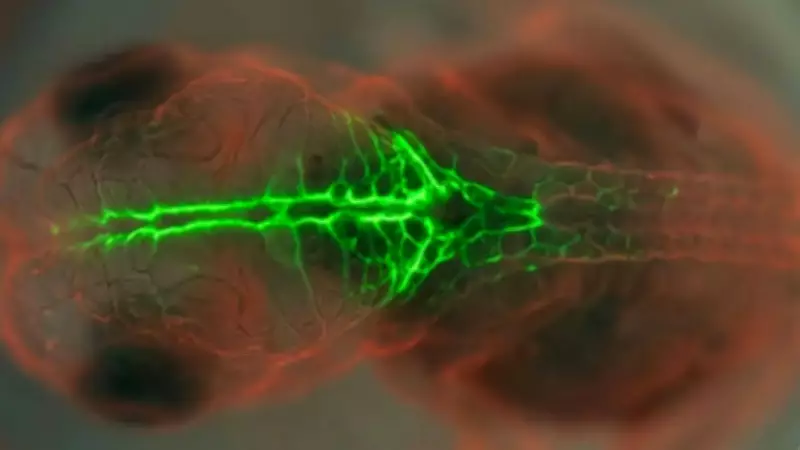
The Role of Fluorescent Quail Embryos in Modern Research
Quail embryos serve as ideal models for studying human neural development because their junctional neural tube region closely resembles human anatomy. By using transgenic quail lines and high-resolution, real-time fluorescent imaging, researchers were able to visualize cellular movements that were previously intractable. As detailed in a University of Queensland thesis, this approach allowed scientists to accurately track midline cells as they migrated and ingressed, providing the first direct evidence of how disruptions in specific proteins cause junctional defects. This method offers a repeatable and observable system for testing potential translational outcomes, bridging basic developmental biology and clinical applications.
Implications for Human Health and Future Screening
Junctional neural tube defects were first formally characterized as a distinct clinical entity in 2017. Unlike more common defects that can be mitigated by folic acid, junctional defects have lacked effective treatment options. By identifying PRICKLE1-mediated EMT as a critical driver of these malformations, scientists now have a specific target for research into preventive medicine. These findings may eventually enable clinicians to develop better genetic screening tools and targeted therapies, potentially reducing the incidence of severe birth defects that currently require complex, lifelong surgical intervention.









