BITS Pilani Hyderabad Research Offers New Hope Against Persistent Bacterial Infections
In the ongoing battle against antibiotic-resistant infections, one of healthcare's most persistent challenges has been the recurrence of infections even after patients complete a full course of antibiotics. A groundbreaking laboratory study conducted at BITS Pilani's Hyderabad campus now presents a promising strategy that could enhance the effectiveness of existing antibiotics against bacteria concealed within protective biofilms.
Understanding the Biofilm Fortress
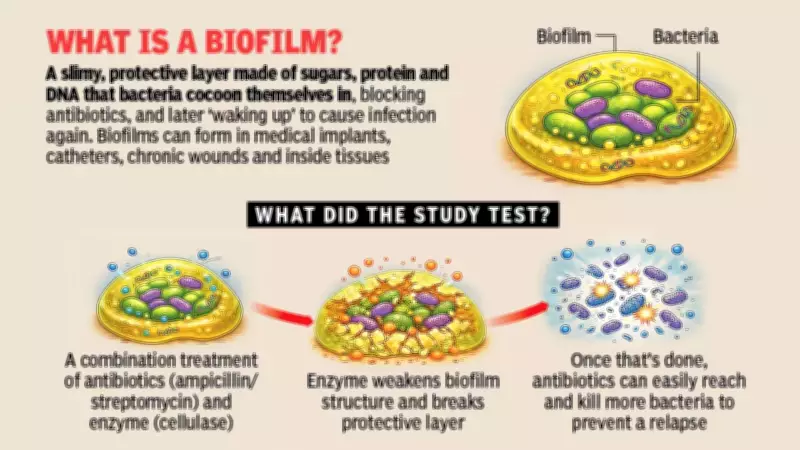
Biofilms represent structured communities of bacteria encased within a self-produced protective layer composed of sugars, proteins, and DNA. This slimy matrix attaches firmly to various surfaces including medical catheters, implants, and chronic wounds, functioning as a formidable shield that effectively blocks antibiotic penetration. Even when antibiotics successfully eliminate the majority of bacterial cells, some enter a dormant state and later reactivate, causing infections to return with renewed vigor.
The study, published in the prestigious Chemical Engineering Journal in March under the title 'Breaching the Biofilm Fortress,' represents a significant shift in research focus. While numerous previous studies concentrated primarily on reducing biofilm mass, this innovative research redirects attention toward preventing the post-treatment resurgence of infections.
Targeting the Protective Matrix Structure
"By targeting the fundamental structure of the biofilm, our approach aims to limit the survival of dormant cells that typically cause relapse," explains study author Ramakrishnan Ganesan from the chemistry department of BITS Pilani's Hyderabad campus. "Our findings strongly emphasize the critical importance of attacking the protective matrix itself rather than merely addressing the bacterial cells within."
To ensure their research reflected real-world conditions, the research team isolated a naturally occurring, biofilm-forming E. coli strain from the vicinity of Patancheru Lake on Hyderabad's outskirts. This area experiences significant exposure to diverse industrial and pharmaceutical effluents, making it an ideal source for environmentally relevant bacterial samples. Genetic analysis revealed the strain to be closely related to sequence type 10 (ST10), a bacterial lineage known to include both harmless and disease-causing variants.
Combination Therapy Shows Remarkable Results
The research team evaluated two widely prescribed antibiotics—ampicillin and streptomycin—both independently and in combination with cellulase, an enzyme specifically capable of breaking down polysaccharides within the biofilm matrix. The underlying concept proved elegantly simple: first weaken the protective barrier, then allow antibiotics to penetrate more effectively to reach their bacterial targets.
Across multiple repeated treatment cycles, the combination therapy demonstrated significantly superior performance compared to antibiotics administered alone. Among all tested treatment combinations, the streptomycin-cellulase pairing delivered the most impressive results, achieving near-complete elimination of viable biofilm cells under controlled laboratory conditions.
Scientific Validation and Safety Assessment
Advanced imaging techniques confirmed extensive breakdown of the protective matrix following cellulase treatment, facilitating substantially improved drug penetration into previously inaccessible biofilm regions. Comprehensive safety tests conducted on mammalian cell lines revealed over 90% cell viability, suggesting excellent biocompatibility at this experimental stage.
"Suppressing biofilm resuscitation—rather than merely reducing biomass alone—represents the key to overcoming antibiotic treatment failure," states the published study. "This approach addresses the fundamental mechanism that allows infections to persist and return despite conventional antibiotic therapy."
Clinical Implications and Future Directions
Jayati Ray Dutta, a professor in the biological sciences department and co-researcher on the study, highlights the practical implications: "Our research suggests that strategically breaking down the biofilm barrier, rather than relying exclusively on increased antibiotic doses, could provide healthcare professionals with an additional therapeutic strategy to manage persistent infections in clinical settings."
The researchers emphasize that while these laboratory findings show tremendous promise, additional comprehensive studies will be necessary before this innovative approach can progress toward clinical application. Future research will need to address scalability, dosage optimization, and potential side effects in more complex biological systems.
This pioneering work from BITS Pilani Hyderabad represents a significant step forward in the global fight against antibiotic-resistant infections, offering hope for more effective treatment strategies that could potentially save countless lives and reduce healthcare burdens worldwide.









